Key Features
10 minutes per day procedure, non-invasive, no drug treatment
Medically Proven, TGA Listed
Product information
MARODYNE LiV
Marodyne LiV is a bone growth and muscle stimulation device specifically developed to increase bone density and quality, while also activating and conditioning the leg muscles in a safe and effective manner.
A very precise low level vibration is transmitted at a high frequency to the body while standing on the device. Only 10 minutes a day is sufficient to make your bones and muscles stronger, and this level of vibration is safe.
Additionally to meeting all vibrational safety limits, full device function and the results it is able to achieve have been medically certified according to the European Union Medically Device Directive.
People with osteoporosis or decreased bone density (osteopenia), or those with reduced muscle strength, balance problems and risk of falling can easily, safely and effectively overcome these conditions by utilising Marodyne LiV for 10 minutes per day. This preventive, natural way of bone and muscle stimulation can help you to live a longer, healthier and independent life.
Technology
What is Low-intensity Vibration?
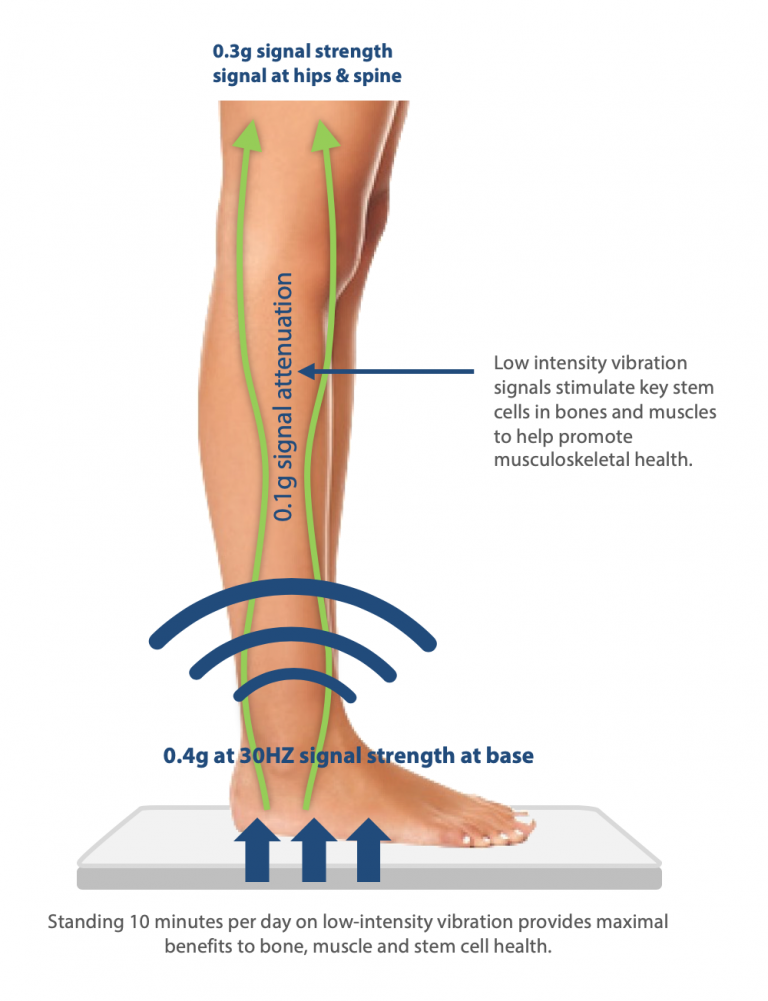
Achieving peak bone mass is a critical determinant of life-long skeletal health. Our bone mass develops from birth to around the age of 30; after that, it slowly begins to decline. In those with osteoporosis, their bones become fragile, which makes them likely to fracture or break. The bones in our body respond to both large low-frequency and small high-frequency forces. By stimulating the body’s stem cells – specifically, those in the legs – Marodyne LiV encourages bone growth and maintains bone health. Marodyne LiV provides an exact low-level vibration (0.4g) transmitted at a high frequency (30Hz) to the person standing on the device. An impulse of 0.4 g is applied to the base (sole of the foot), approx. 0.1 g is damped in the leg area and 0.3 g is passed on to the hip and spine (‚g‘ stands for gravity – our acceleration due to gravity with ~ 9.81 m/s²).
Marodyne LiV impulses correspond to natural, mechanical movement impulses – i.e. impulses that are picked up on the skeleton step by step, for example, when descending a staircase. The adjustment is automatic, individually for each user -this weight measurement and adjustment to each user, controlled by a microprocessor, ensures safety. This stimulates osteoblast (bone-building) activity while inhibiting osteoclast (bone wasting) activity. Studies have established that just 10 minutes of LiV a day can help to keep bones strong and healthy.
Product Features
Marodyne LiV is designed for years of use and has a sturdy metal construction to withstand the usual daily use of the device. A visual display shows the session time and the position of the user for optimal exposure to LiV during the session.
Marodyne LiV is portable and is supplied with a smart travel bag to enable the user to take their device with them and continue their treatment wherever they are.
Each device consists of the following components:
– Marodyne LiV device
– power supply with cord
– instructions for use
– power cord, length < 3m
– travel bag

System weight: 8.1 kg (17.85 lb)
System Dimensions
H/W/D: 7.7 cm (3.03 in) / 45.7 cm (17.99 in) / 35.6 cm (14.01 in)
Low intensity vibrations: approx. 30 Hz / body acceleration: 0.4 g ± 20% / amplitude: 50 to 200 microns
Power Supply
Input voltage: 100 – 240 Volts AC / input frequency: 50 – 60 Hz
Output voltage: 16 Volts DC / output amperage: 4.38 AMPS, 70 W max.
Protection: Class 1
Product Benefits
Safe and effective
Marodyne LiV is a medical device (Class 2a) and certified and approved by the European Union (MDD 93/42 / EEC Annex II). It’s the world’s only clinically-approved, non-drug treatment. The device is TGA listed, see link here.
By using Marodyne LiV for just 10 minutes a day you can:
- maintain and increase bone mineral density (BMD).
- build muscle strength and muscle mass in the legs.
- stimulate blood circulation and lymphatic flow.
- improve balance, postural reflexes (control) and co-ordination.
- stimulate your metabolism to boost bones and muscles.
- support postural stability, balance and coordination – assists in falls prevention.
- reassign stem cell, (MSC’s) to bone cell formation – creating healthier bones
- suppress stem cell formation of new fat.
Marodyne LiV is tested to deliver benefits to:
- older patients with osteoporosis
- post-menopausal women with poor bone health
- people at risk-of developing osteoporosis
- children and young people with bone-damaging diseases and conditions
- medical professionals who want to reduce falls
Treatment benefits
Marodyne LiV has significant benefits over currently available treatments prescribed to patients. Low-intensity vibration is:
- totally drug-free
- uses the body’s natural signals to stimulate bone growth
- has no side effects
- simple to use at home without supervision
- highly effective
- non-invasive
- proven to reduce falls in older people
Marodyne LiV is already transforming the lives of those with poor bone health – helping them to be happier, healthier, and more independent.
Backed by science
Marodyne LiV has been developed by a team of scientists, researchers, developers, and manufacturers of medical products. The team is headed by Professor Dr. Clinton Rubin, one of the world’s foremost biomedical engineers.
The technology inside Marodyne LiV was developed in collaboration with NASA and the National Institutes of Health (NIH) and represents almost 40 years of research and $70m of investment.
The development and continued improvement of Marodyne LiV is supervised by a scientific advisory board with leading experts from the fields of bio medicine, orthopaedics, osteoporosis, metabolism, rehabilitation medicine and stem cell research.
Company profile
BTT Health
BTT Health is the manufacturer of the ‘Marodyne LiV’ – Low-intensity vibration therapy platform as well as the ultrasound bone growth stimulator ‘bone4ce’. BTT Health GmbH is a research and development company, specializing in safe intervention for illness, disease and injury.
Headquartered in Inning am Ammersee close to famous Munich/Bavaria in Germany, BTT Health’s medical leadership team includes experts in the fields of biophysical stimulation, chronic disease and injury, and medical device research, development, and manufacturing.


